LITE Liner® Silicone

The connection between residual limb and socket is the key for a real high-quality fitting of a prosthesis.
The wide range of silicone liners eased it to find the best solution for your patient upon the indication for the different needs.
LITE Liner® Silicone features a high stability and a moderate damping. They are available with or without distal connector and in different sizes and colours.
Selection matrix
| Fabric cover: |
4W1
4W4
4U4 4L4
|
LITE Liner® Clear LITE Liner® Skin Tex LITE Liner® Skin Tex U LITE Liner® Skin Tex L |
| Style: |
0
1
|
Locking Cushion |
| Material: |
21
22
34
62
|
approx. 2,5 mm DuraSil-Silicone, hardness: normal approx. 2,5 mm ComfortSil-Silicone gel, hardness: soft POST-OP Liner, very soft, no fabric cover approx. 6 mm ComfortSil-Silicone gel, hardness: soft |
| Matrix: |
00
05
10
15
TF
|
no Matrix approx. 5 cm Matrix approx. 10 cm Matrix approx. 15 cm Matrix total length Matrix (for AK), thickness approx. 2,5 mm |
| Size: |
16-45
16-55
26,5-55 |
LITE Liner® Clear LITE Liner® SkinTex LITE Liner® SkinTex TF |
| Color: |
-G - |
light grey (LITE Liner® SkinTex) clear (only LITE Liner® Clear) |
| Ordering example: 4W4022-10-28-G = LITE Liner® Skin Tex 2 soft, 10 cm Matrix, size 28, Color light grey | ||
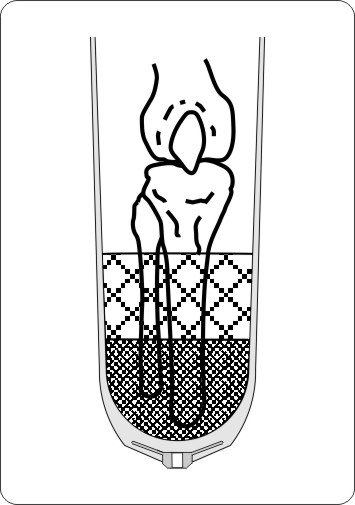
Determination of the matrix length:
For different lengths of the residual limb, the length of the matrix (reinforcement) can be chosen from 0, 5, 10 and 15 cm. The matrix should cover the tibia but should not be extended above the level of the fibula head. This ensures a higher stability of the soft tissue.

Determination of the size:
To determine the correct liner size, the circumference of the residual limb is measured 4 cm above the distal end. Select that size being exactly the same of the stump circumference or the next smaller one.
Stump paddings:
If the profile of the residual limb is to be compensated, the Distal Cup is recommended for conical stumps making it more cylindrical. The size of the silicone liner is then to be determined above the donned Distal Cup.
In case of a flat or square formed residual limb, the Silicone Pad can be used to replace missing tissue and to achieve an optimum contact between skin and silicone.
The silicones used have been tested for biocompatibility according to DIN EN ISO 10993-5 and 10993-10. The certificates can be delivered anytime on request.


